Bicon SynthoGraft Summary of Safety and Clinical Performance Document number: SSCP-002 Document revision: 01 Date issued: April

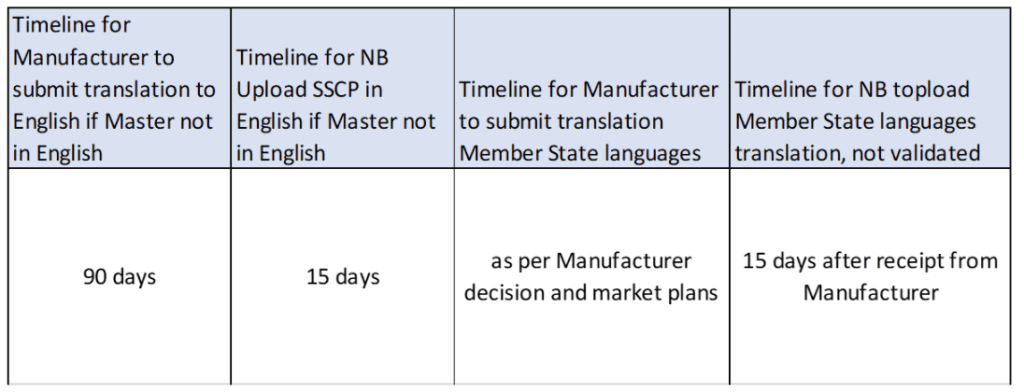
Summary of Safety and Clinical Performance (SSCP): 5 Challenges to be met - GMED Medical Device Certification
How the new European regulation on medical devices will affect innovation | Nature Biomedical Engineering

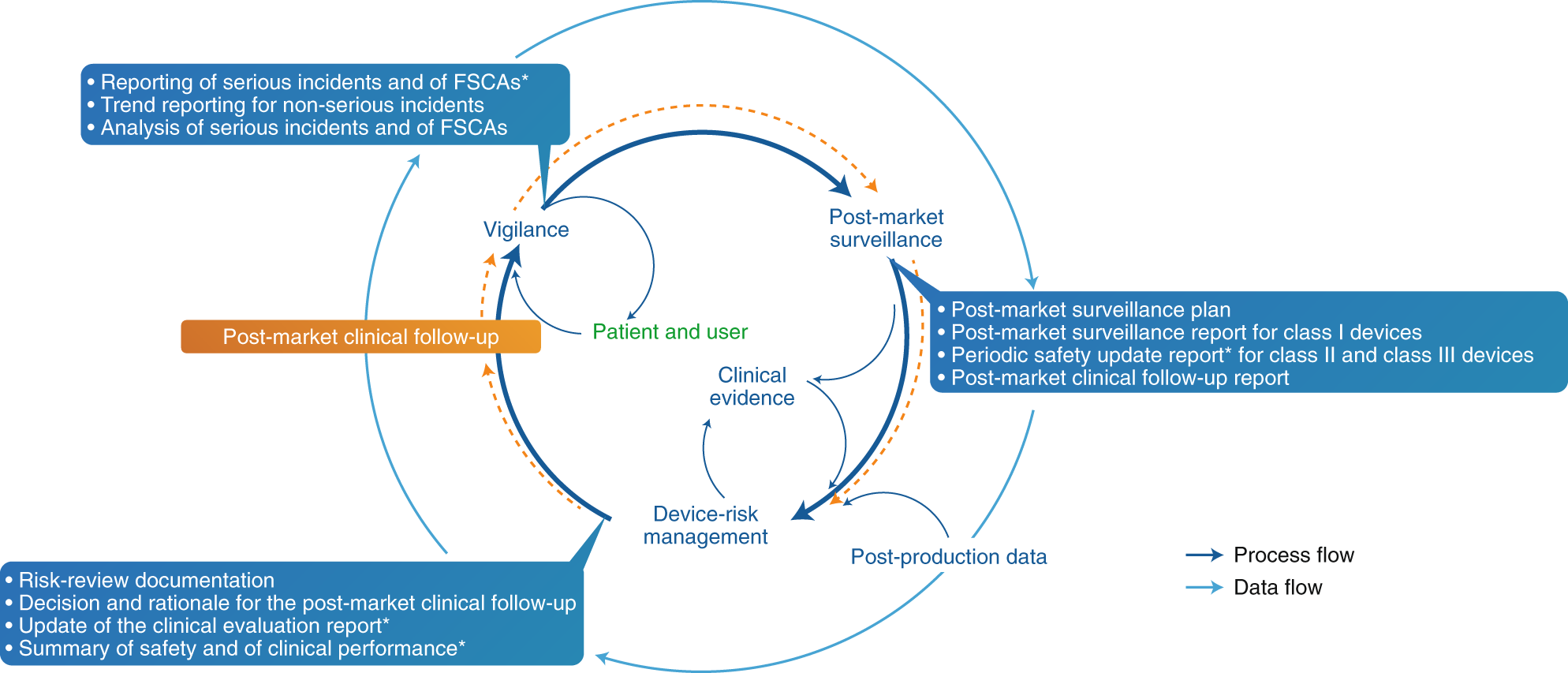
The Summary of Safety and Clinical Performance (SSCP) Provides Valuable Information to Health Care Providers, Patients, and Manufacturers - Criterion Edge

Summary of Safety and Clinical Performance (SSCP): 5 Challenges to be met - GMED Medical Device Certification

![Summary of Safety and Clinical Performance [ISO 13485 templates] Summary of Safety and Clinical Performance [ISO 13485 templates]](https://advisera.com/wp-content/uploads//sites/14/2021/08/24.3_Summary_of_Safety_and_Clinical_Performance_Integrated_Preview_EN.png)